Im not sure why thats such a prominent myth.
Note this will be some reading
Both thc and butter obviously being highly lipophilic..,,
Wait when you say thick skull, are you wanting the simplest explanation possible?
Back to ^.... both being fat loving or non polar they mix together fine..homogenously.
Theres no reason for an emulsifier
And in fact..
"Hi guys. I'm new and need some help. I've made canna butter and canna oil from canola oil. This is my first attempt with Cocunut oil. I made it in a crockpot I used lecithin and let it cool, then placed it in the freezer. After getting rid of the water and plant material I put it in the pantry in the Cocunut oil container. It seems to have separated again with the top half a dark clear oily liquid and the bottom half a split pea colored oily liquid. Which should I use, or is it ok to stir it up and use it all?"
From like a week ago edit jan 25th
The lecithin pulled in excess water which seperated once the emulsion broke down...
As to why its counterproductive and not just a possible hassle...
Ok read and bare with me. Ive made additons to the quote. What you dont understand ask away but keep reading i may clarify
Essentially both will work, but thc is too non polar to be effectively absorbed and hiding it in a bubble stops you from using your butter as a medium to lower polarity and enhance efficiency
- qwizokingWell-Known Member
i have to post this quite often as im asked all the time
forgive grammar btw and choppy structure. any questions or advice on simplification go for it
"In the pharmaceutical industry, lecithin acts as a wetting, stabilizing agent and a choline enrichment carrier, helps in emulsifications and encapsulation, and is a good dispersing agent. It can be used in manufacture of intravenous fatinfusions and for therapeutic use.
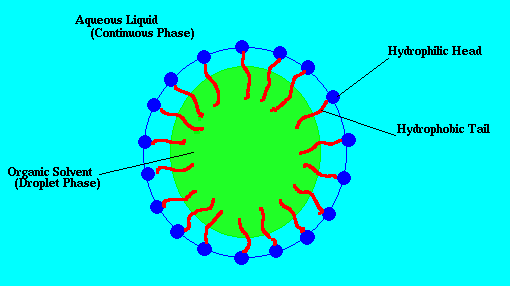
So lecithin
is an excellent emulsifier. In aqueous solution, its phospholipids can form either liposomes, bilayer sheets, micelles, or lamellarstructures,depending on hydration andtemperature.
A number of different chemical and physical processes and mechanisms can be involved in the process of emulsification
, emulsification takes place by reduction of interfacial tensionbetween two phases
the emulsifying agent creates a film over one phase that forms globules, which repel each other. This repulsive force causes them to remain suspended in the dispersion medium
An appropriate "surface active agent" (or "surfactant") can increase thekinetic stability of an emulsion so that the size of the droplets does not change significantly with time. It is then said to be stable.
Thats essentially what an emulsion is
In the quote you can see how their product seperated..
-
this is the droplet
Its attempting to divide the substances. Both being fatty it has trouble but does
sorry not in an explaining kind ofmood today ^from wiki
now i assume you all know why this is not advantageous. think why we use coconut oil in the first place...or butter lol
If not check my coconut oil post..
Given.my bakground i want everything i do to be optimum. Lecithin use will still work
You can just eat decarbed thc and get high...but
we put it in a carrier..a fat... to lower polarity and increase efficiency..
thc has a logp of about 7
In practiceoptimum colonic absorption. is 1.32, intestinal is 1.35, oral 1.8, Cns 2, percutaneous at 2.6 and sublingual 5.5.
very important in roa and pharmacology
Lauric acid having a log p of about 4.5..and smallest mct having a logp about 1.8...
lauric acid is the star component of coconut oil.
- hiding thc inside a bubble as pictured is counter productive.
again we want the thc mixed homogenous like in the solution to make use of the carrier and polarity difference.
- This gives a faster onset and higher bioavailability
we use coconut oil only for oral use however as it has a fairly high log p, and is still an mct, which gets taken up differently, important . as well as holding most insolution given longer chain length meaning more non polar
have you read any of my posts on mct and coconut oil?