I guess all labs have their good days and bad days ... well, (sadly), amateurs have more than their fare share of bad days


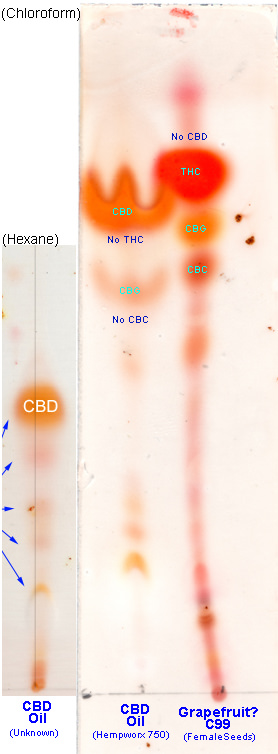
I've just done another TLC today, this time on a CBD oil, as well as C99 by FemaleSeeds Co (perhaps the grapefruit pheno or just another pheno thats popped out from whatever theyve crossed C99 with... def not the pineapple/tropical/classic C99 tho, but very much still in the neighborhood).
It's not my cleanest TLC though! As I was only doing 2 samples I decided to cut the silica plate in half so I could do two samples per halfplate, each in a different eluent for the mobile phase.
But, unfortunately there was some minor interaction between the lanes. From now on, I think I will only cut a plate in half when doing 1 sample (1 lane per halfplate) in 2 eluents. The aluminium silica plates are only about $1-2 each anyway, and TLC isn't something I do on a regular basis anyway, so I should stop being a tightass with those!
A little ant also ran across the silica plate, probably leaving a pheromone trail, the little ****! I highly doubt it would affect the TLC in any noticeable way though, but I wouldn't want to bet on it, especially as this was one of my worse TLC runs, although that could just be a coincidence.
Anyway here is the first halfplate (the two lanes on the RIGHT), which I developed with Chloroform as the mobile phase eluent. On the LEFT, the smaller one is just a copy from an image ive already posted here, which was the only other time I TLC'd a CBD oil sample (pretty much all my other testing has been with buds). I'm not sure why the Grapefruit lane isn't straight, perhaps there was a slight bend in the plate being only a half-plate, but I haven't seen that problem before with other halfplates, and even if it was bent you'd think it would still go upright, so perhaps it was just slightly oriented off-center due to being in a cylindrical jar with a raised bottom which might've offset it ever so slightly)
I've also TLC'd an official Grimm Brothers C99, so for comparison you can see that in the last image in [
this post] a bit earlier in this thread.
ps. you can see two dark dots to the right of the Grapefruit lane's CBG dot ...they're clumps of Fast Blue BB (i wasn't patient enough with the coffee filter paper and pushed a bit too hard, so some FBBB ended up in the mix, whoops... be very gentle when squeezing the FBBB through the coffee filter paper)
While I can't remember what the Unknown CBD oil i tested is called, and although I only used Hexane : Diethyl Ether as the eluent, you can see that its profile is very similar to the CBD oil I tested today (Hempworx 750). Note: This is not an endorsement, and I'm not saying you should or shouldn't get it.
There's a lot of good/legit CBD oils out there, but sadly a lot of scamming going on, and that's ****ing with peoples lives, and in the case of cancer patients they simply don't have time, OR money, OR health & wellbeing to lose to scammers. Always do a lot of googling about a CBD oil before you buy it - do some basic background checks on the company, and search the web to see if other people have found it helpful.
This particular CBD oil, Hempworx 750, purports to contain 750mg of CBD (in a 30ml/1oz bottle, so that would be 2.5% CBD, W/V%), and comes infused in hemp seed oil, and peppermint oil for taste. The word from my friend going through cancer who's just tried it said it tasted good, which might be good for people who don't like that
planty taste some oils have. Or simply add a drop or two of your own peppermint oil to whatever CBD oil you have, no rocket science there!
I've never seen that "W" shape/effect that can be seen in the CBD oil's CBD spot on the TLC plate ... I'm wondering if that's a component of the peppermint oil just ahead of it that might've caused that (clearly it has SOME other molecular components ahead of it near the top, as you can see how much lower on the plate the CBD dot is... CBD appears
above THC, so you can tell where the Grapefruit's CBD dot would exist if it had one). It'd be great to have another try on a full plate etc, but the stuff is expensive and comes in tiny bottles. What's the dark-orange line on the top of the CBD spot? I'm not sure - perhaps its due to the minor interaction with the red THC dot in the lane beside it, because i've never seen such a 'line' before... always DOTS. However, perhaps the molecule dot above it is 'restrictive' in a way, something like a wall, which might lead to a buildup along the edges and therefore resulting in a darker color from the higher density, but this is pure speculation and guessing at possibilities.
So, just judging from those two hemp-derived CBD oils (granted, not a big sample size), my takeaway is this:
- CBD oils are a great way to LOAD UP on CBD, without having to worry about THC taking over. As CBD isn't psychoactive obviously we can handle a LOT of CBD, but we can only handle so much THC before we fall asleep, and that's a significant limiting factor. I now I'm not saying anything new there, but is an important aspect for patients. Some medical trials with CBD have used very high levels of CBD, and you simply can't obtain those levels with for example "1:1" CBD:THC strains.
- However, a lot of trials also show the SYNERGY that CBD and THC have - THC often seems to positively increase CBD's effects (something the cannabis community has known for a long time, and is now finally getting scientifically affirmed). So, after your medicinal CBD oil drops, perhaps have "a quarter of a cookie" or something to introduce a small but manageable amount of THC. (Or the whole cookie, or four, whatever)

Anyway, so that was the plate developed with just pure Chloroform as the mobile phase eluent ...
but I also use Hexane : Diethyl Ether (4:1) ... I'm still not sure which eluent I prefer (they both seem to have pros and cons, but also
complement each other nicely), and I've had nothing but success with it so far ...
... but NOT TODAY! Just one of those days unfortunately! ...
WHOA! Massive fail ..... the
extract didn't even get out of the starting blocks!!! (even though the
eluent crossed the finishing line!)
The only saving grace I guess, is that yes we can see the CBD Oil lane is clearly showing a fat orange CBD dot, while the C99 lane is clearly showing a fat red THC dot... it's not much information, but it's better than nothing and still definitely provides a
slightly clearer indication than
Beam's Test, as Fast Blue BB is far more specific in indication of cannabinoids. But, for whatever reason, it failed to SEPARATE the substance into its lesser molecules, so we've basically put a bread in the oven and it's failed to rise due to some problem probably with the yeast.
In case you're wondering, yes the eluent did capillary-action all the way to the top (and you can see some minor staining at the top), so that's not the problem.
THE PROBLEM:
- The extract failed to be moved up the plate via capillary action.
So, what's responsible for driving it up? there are two fundamental parts...
- first we use Hexane to create the EXTRACT... we need to extract the cannabinoids from the target substance (Hexane is something of a molecular magnet)
- then we use the eluent (eg. Chloroform, or Hexane : Diethyl Ether) for the MOBILE PHASE, where capillary action drives the eluent up to the top of the plate, separating molecules into groups in the process
I believe I've made a mistake in either of these two.
POSSIBLE CAUSES: (the only ones i can think of - it's a fairly straightforward procedure afterall)
- MY BET: perhaps my hexane : diethyl ether measurement was slightly off (i don't know how sensitive the ratio is). In one of my first posts I showed experiments with different eluents that help show how important it is to use "the right eluent for the job (and there aren't many for this)", but I'm confident I was within 1ml of 8ml of hexane, and pretty sure I was close to the 2ml for diethyl ether (should be 4:1 ratio). I've never had a problem with it before, but measuring this time was trickier as my syringes now no longer reach into the ever-depleting level of eluent in the bottle, so I was happy at the time to be less precise. However, there is obviously a reason why the two are used together, and there's obviously a reason for the 4:1 ratio ... so, perhaps there is no elbow room there. Perhaps we need to be very precise with this mix. But in my previous experiments with different eluents, there was never this "gridlock" problem! Even water had no problem driving forward, albeit without separation. So, if this is the problem, the solution is of course simple: be precise ... it's not difficult, especially now in retrospect haha
- UNLIKELY 2ND BET: perhaps I didn't use enough hexane in the extraction phase (possibly resulting in extract which is too 'sticky'), but it didn't affect the Chloroform plate.
- UNLIKELY: I actually used 4 drops per lane, usually I just use 3, but surely that 1 extra drop couldnt have this much effect, especially as it didn't affect the Chloroform plate.
- UNLIKELY: surely that ant that walked across the plate didn't leave a pheromone trail that could have this much effect, especially as it didn't affect the Chloroform plate.
I might be able to revisit this in the future to try to set up a couple of experiments which can test some of these possibilities to try to rule them out.
Due to lack of publicly available knowledge about this, I basically have no other option but to keep experimenting. Hopefully some of you will too